12th Standard Chemistry / வேதியியல் Subjects Question Paper Software Subscription
QB365 covers complete information about Tamilnadu 12th Standard 2022-2023 Chemistry / வேதியியல் Subject. Question Bank includes 12th Standard 2022-2023 Chemistry / வேதியியல்'s Book back questions, other important questions, Creative questions, Extra questions, PTA quesions, Previous Year questions and other key points also. All question with detailed answers are readily available for preparting Chemistry / வேதியியல் question papers.
All Chapters Covered

Create Unlimited Question Papers

Access anywhere anytime

Multiple Pattern Question Papers
Share your Question Paper

Font size, line spacing, watermark etc,
Our Other Subjects 12th Standard
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
Atomic Structure - II
Periodic Classification - II
p - Block Elements - II
d- Block Elements
f- Block Elements
Coordination Compounds and Bio-Coordination Compounds
Nuclear Chemistry
Solid State - II
Thermodynamics = II
Chemical Equilibrium - II
Chemical Kinetics II
Surface Chemistry
Electro Chemistry I
Electro Chemistry II
Isomerism in Organic Chemistry
Hydroxy Derivatives
Ethers
Carbonyl Compounds
Carboxylic Acids
Organic Nitrogen Compounds
Biomolecules
Chemistry in Action
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
Atomic Structure - II
Periodic Classification - II
p - Block Elements - II
d- Block Elements
f- Block Elements
Coordination Compounds and Bio-Coordination Compounds
Nuclear Chemistry
Solid State - II
Thermodynamics = II
Chemical Equilibrium - II
Chemical Kinetics II
Surface Chemistry
Electro Chemistry I
Electro Chemistry II
Isomerism in Organic Chemistry
Hydroxy Derivatives
Ethers
Carbonyl Compounds
Carboxylic Acids
Organic Nitrogen Compounds
Biomolecules
Chemistry in Action
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023 Syllabus
Atomic Structure - II
Dual properties of electrons - de-Broglie relation - Heisenberg’s uncertainty principle - Wave nature of an electron - Schrodinger wave equation (only equation, no derivation) - Eigen values and Eigen function- significance only - molecular orbital method. Application to Homo diatomic and Hetero diatomic molecules - Metallic Bond - Hybridization of atomic orbitals Hybridization involving s, p and d Orbitals - Types of forces between molecules.
Periodic classification - II
Review of periodic properties - Calculation of atomic radii - Calculation of ionic radii - Method of determination of Ionisation potential - Factors affecting ionisation potential - Method to determine the electron affinity - Factors affecting EA - Various scales on electro negativity values.
p - Block Elements - II
Group -13 General trends - Potash alum- Preparation, Properties and uses - Group 14 General trends - Silicates - Types and structure - Silicones - Structure and uses - Extraction of lead - Group - 15. General trends - Phosphorous - Allotropes and extraction - Compounds of phosphorous - Group - 16. General trends - H2 SO4 - Manufacture and properties. - Group - 17 General characteristics. Physical and Chemical properties - Isolation of fluorine and its properties - Interhalogen compounds Group-18 Inert gases - Isolation, properties and uses.
d - Block Elements
General characteristics of d-block elements - First transition series - Occurrence and principles of extraction - chromium, copper and zinc - Alloys - Second transition series - Occurrence and principles of extraction of silver - Third transition series - Compounds - K2Cr2O7, CuSO4 5H2O, AgNO3, Hg2Cl2, ZnCO3, Purple of Cassius.
f - block elements
General characteristics of f - block elements and extraction - Comparison of Lanthanides and Actinides - Uses of lanthanides and actinides.
Coordination Compounds and Bio-coordination
Compounds An introduction - Terminology in coordination chemistry - IUPAC nomenclature of mononuclear coordination compounds - Isomerism in coordination compounds - Structural isomerism - Geometrical isomerism in 4 - coordinate, 6 - coordinate complexes - Theories on coordination compounds - Werner’s theory (brief) - Valence Bond theory - Crystal field theory - Uses of coordination compounds - Bio coordination compounds. Hemoglobin and chlorophyll. Unit 7 - Nuclear chemistry Nuclear energy nuclear fission and fusion - Radio carbon dating - Nuclear reaction in sun - Uses of radioactive isotopes.
Solid state II
Types of packing in crystals - X-Ray crystal structure - Types of ionic crystals - Imperfections in solids - Properties of crystalline solids - Amorphous solid.
Thermodynamics - II
Review of I law - Need for the II law of thermodynamics - Spontaneous and non spontaneous processes - Entropy - Gibb’s free energy - Free energy change and chemical equilibrium - Third law of thermodynamics.
Chemical equilibrium II
Applications of law of mass action - Le Chatlier’s principle.
Chemical Kinetics -II
First order reaction and pseudo first order reaction - Experimental determination of first order reaction - method of determining order of reaction - temperature dependence of rate constant - Simple and complex reactions.
Surface Chemistry
Adsorption - Catalysis - Theory of catalysis - Colloids - Preparation of colloids - Properties of colloids - Emulsions
Electrochemistry - I
Conductors, insulators and semi conductors - Theory of electrical conductance - Theory of strong electrolytes - Faraday’s laws of electrolysis - Specific resistance, specific conductance, equivalent and molar conductance - Variation of conductance with dilution - Kohlraush’s law - Ionic product of water, pH and pOH - Buffer solutions - Use of pH values.
Electrochemistry - II
Cells - Electrodes and electrode potentials - Construction of cell and EMF - Corrosion and its preventions - commercial production of chemicals - Fuel cells.
Isomerism in Organic Chemistry
Geometrical isomerism - Conformations of cyclic compounds - Optical isomerism - Optical activity - Chirality - Compounds containing chiral centres - D-L and R-S notation - Isomerism in benzene
Hydroxy Derivatives
Nomenclature of alcohols - Classification of alcohols - General methods of preparation of primary alcohols - Properties Methods of distinction between three classes of alcohols 1°, 2° and 3°) - Methods of preparation of dihydric alcohols. (glycol) - Properties - Uses - Methods of preparation of trihydric alcohols - Properties - Uses - Aromatic alcohols - Methods of preparation of benzyl alcohol - Properties - Uses - Phenols - Manufacture of phenols - Properties - Chemical properties - Uses of Phenols.
Ethers
Ethers - General methods of preparation of aliphatic ethers - Properties - Uses - Aromatic ethers - Preparation of anisole - Reactions of anisole - Uses.
Carbonyl Compounds
Nomenclature of carbonyl compounds - Comparison of aldehydes and ketones - General methods of preparation of aldehydes - Properties - Uses Aromatic aldehydes - Preparation of benzaldehyde - Properties - Uses - Ketones - general methods of preparation of aliphatic ketones (acetone) - Properties - Uses - Aromatic ketones - preparation of acetophenone- Properties - Uses - preparation of benzophenone - Properties.
Carboxylic Acids
Nomenclature - Preparation of aliphatic monocarboxyli c acids - formic acid - Properties - Uses - Tests for carboxylic acid - Monohydroxy mono carboxylic acids - Lactic acid - Sources - Synthesis of lactic acid - Aliphatic dicarboxylic acids - preparation of dicarboxylic acids - oxalic and succinic acids - Properties - Strengths of carboxylic acids - Aromatic acids - Preparation of benzoic acid - Properties - Uses - Preparation of salicylic acid - Properties - Uses - Derivatives of carboxylic acids - Preparation of acid chloride - acetyl chloride (CH3COCl) - Preparation - Properties - Uses - Preparation of acetamide - Properties - Preparation of acetic anhydride - Properties - Preparation of estersmethyl acetate - Properties.
Organic Nitrogen Compounds
Aliphatic nitro compounds - Preparation of aliphatic nitroalkanes - Properties - Uses - Aromatic nitro compounds - Preparation - Properties - Uses - Distinction between aliphatic and aromatic nitro compounds - Amines - Aliphatic amines - General methods of preparation - Properties - Distinction between 1°, 2°, and 3° amines - Aromatic amines - Synthesis of benzylamine - Properties - Aniline-preparation - Properties - Uses - Distinction between aliphatic and aromatic amines - Aliphatic nitriles - Preparation - properties - Uses - Diazonium salts - Preparation of benzene diazoniumchloride - Properties.
Bio molecules
Carbohydrates - structural elucidation - Disaccharides and polysaccharides - Proteins - Amino acids - structure of proteins - Nucleic acids - Lipids.
Chemistry in Action
Medicinal chemistry - Drug abuse - Dyes - classification and uses - Cosmetics - creams, perfumes, talcum powder and deodorants - chemicals in food - Preservatives artificial sweetening agents, antioxidants and edible colours - Insect repellant - pheromones and sex attractants - Rocket fuels - Types of polymers, preparation and uses.
Nuclear Chemistry
Difference between chemical reactions and nuclear reactions - Type of Nuclear Reaction - Radiocarbon Dating - Nuclear Reactions Taking Place in Sun (Stars) - Uses of Radioactive Isotopes.
Features in Question Paper Preparation software

(or) type Question

Add or Remover

Sub Questions

Adding Notes

Multiple Pattern

All subjects available
How to Create 12th Standard Chemistry English Medium Question Paper
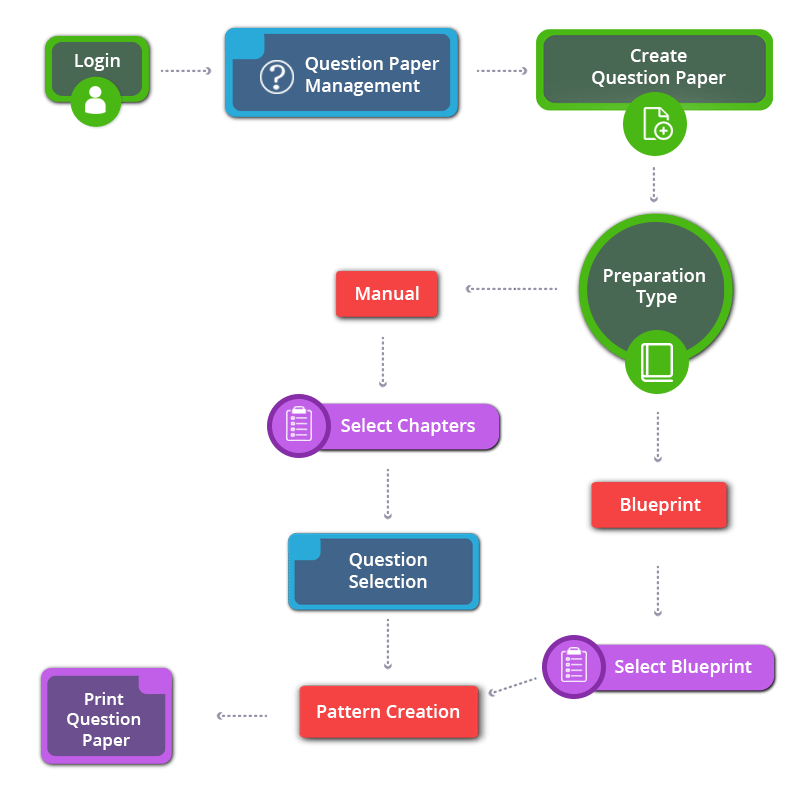

12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
- Covers all chapters
- Unique Creative Questions
- Unlimited Question Paper
- Multiple Patterns & Answer keys
0



